 
Sharing is not so equal and you get polar covalent bonds. In between covalent bonds and ionic bonds where the Or you have covalent bonds where we outright share electrons. Things like ionic bonds where one character swipes an electron from another character and says, "Hey, but now we'reĪttracted to each other," and you get something like salt. That you can view it as something of a spectrum. Well if you were to take aīar of a salt right over here, if you were to try toīend it, it's very rigid. You can imagine these iron ions in this pudding, or this sea of electrons. It conducts electricity because these electrons can Many of the characteristics we associate with metals. And those valence electrons are not fixed to just one of the atoms, Ions with opposite charges will attract one another creating an ionic. Another way to think about it is, is that metals, when theyīond in metallic bonds, they will have overlapping Ionic bonds occur between metals, losing electrons, and nonmetals, gaining electrons. Positive ions are attracted to the sea of negativity, the Electron here that has a negative charge. 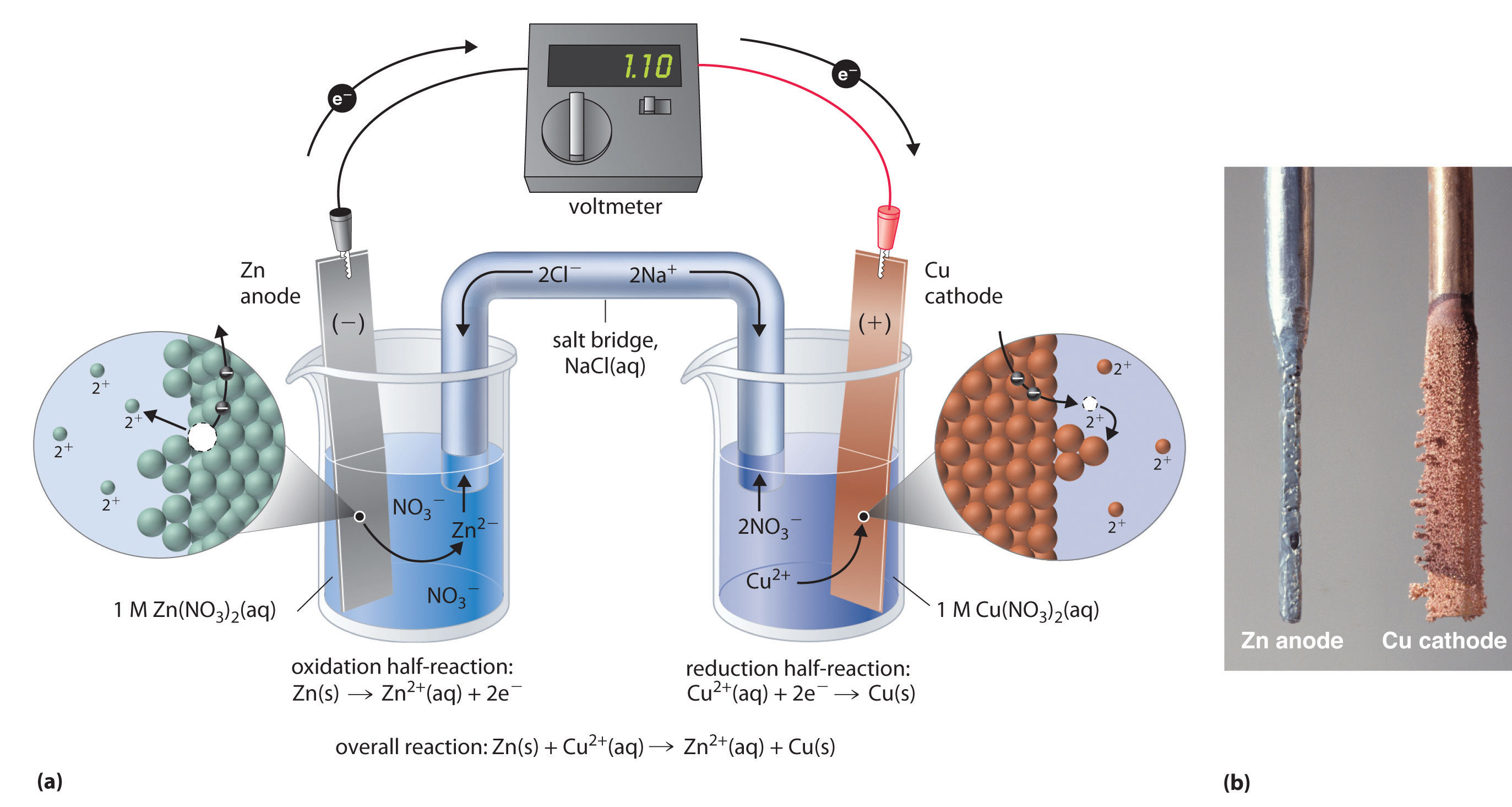
And electrons are not this big, but this is just so that you can see it. Fe plus, and we're imagining that they have this positive charge because they've allĬontributed an electron to this sea of electrons. So what it can look like is, each of the irons lose an electron, I'll draw it a little bit bigger. And what's interestingĪbout metallic bonds, I'll draw it down here, is that metals like to share their electrons with the other metals. But when they are mushed together, they will form a metallic bond. And if they're just atoms by themselves they're going to be neutral. So Fe, Fe, Fe, Fe, hope you can read that. Known as the metallic bond, which I think I know a little bit about because I was the lead singer of a metallic bond in high school. Now the last type of bond I'm going to talk about is This is why metals are more able to lose their electrons in ionic bonds and delocalize their electrons in metallic bonds, since they don't have as strong of a pull on them as non-metals.  
Having more electron shells means that the valence electrons are farther from the nucleus and 'feel' less attraction to the protons. And electronegativity also increases as you move from down to up along a group because the number of electron shells decreases. Having a higher effective clear charge means that negative electrons 'feel' more of the proton's positive charge from the nucleus. Electronegativity increases as you move left to right across a period because the effective nuclear charge increases. The nonmetals occupy the upper-right corner of the periodic table where electronegativity values are the highest. Electronegativity essentially being a measure of how well atoms can hold onto electrons. The giant structure of metals and metallic bonding explain their properties.The main difference between the two groups on the atomic level is that nonmetals are more electronegative than metals. This is why metals are good conductors of electricity. If a potential difference is applied across a piece of metal, the electrons will move, carrying an electrical current. The force of this attraction is very strong. The electrostatic attraction between these opposite charges creates the metallic bonds. This model represents metal crystals as being made up of positive metal ions surrounded by a sea of negative electrons. A more complex model is needed to explain the bonding in metals. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
